Glycans and Influenza

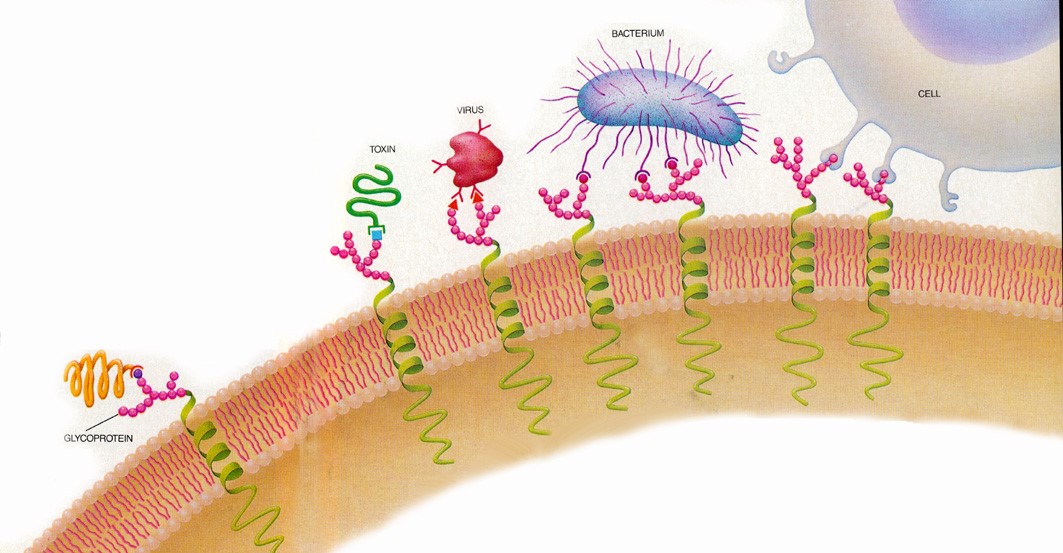
Viral infection typically begins by one or more of viral glycan-binding proteins (lectins) recognizing and binding the glycans on the surface of our cells.
Viral infection typically begins by one or more of viral glycan-binding proteins (lectins) recognizing and binding the glycans on the surface of our cells.
The first identified microbial lectin was the hemagglutinin from the influenza virus. A glycoprotein itself, its name originates from the ability to crosslink red blood cells by binding to terminal sialic acids on their surface. Sialic acids are a family of monosaccharides, and one of glycan components. Influenza hemagglutinin is one of the best-studied lectins and its specific recognition of distinct glycan structures, namely the type of covalent bond between 2 monosaccharide units (sialic acid-2,3-galactose, vs. sialic acid-2,6-galactose) on the surface of host target cells is the main barrier for influenza viruses to cross between species. The importance of this feature became apparent during the spreading of the most pathogenic bird influenza virus H5N1, raising global public concern over a possible human pandemic threat. A single amino acid substitution altered the specificity of H5N1 viruses, enabling them to infect human cells.
The H5N1 virus belongs to the group A of influenza viruses, the only group of influenza viruses known to cause flu pandemics. Interestingly, both letters in the name of the influenza A viruses are related to glycans, thus underlining the importance of glycans in viral infection. The H stands for the aforementioned hemagglutinin, necessary in the initial stages of viral infection, while the N stands for neuraminidase, i.e. sialidase, necessary for the release of viral progeny from infected cells, but also recently discovered to play an important role during infection.
The inhibition of pathogen binding to our cells is an important element of protection against viruses. Therefore, the two major surface glycoproteins on the surface of influenza virus – hamagglutinin and neuraminidase, are the primary target of neutralizing antibodies produced by our immune system during immunization, either by infection or by vaccination. These two viral glycoproteins are also targets for other, non-antibody types of anti-viral molecules.

Glycan science and biological age
Go deeper with GlycanAge for research and cohort studies, peer-reviewed GlycanAge research case studies, and methodology behind glycan-based biological age testing.

The Future of Healthcare is Preventative, Personalised, and Powered by Glycans
Whether you’re improving your own health, supporting patients, or driving research, GlycanAge helps you turn science into action.
O-linked Glycosylation Explained
The blueprint for how proteins are formed is encoded in our DNA, but that’s only part of the story. Once a protein is built, it can undergo further modifications through the attachment of various molecules, altering its structure and function. These changes, collectively known as post-translational modifications, play…
August 27, 2024
10 minutes

Glycosylation: Impact on Health and Disease
Glycans are essential for all life. These are long-chain sugars that are present on proteins, lipids (also known as fats) and nucleic acids, and which make these biomolecules functional. When we think of sugars, our first thought may be about the sugars from food we eat, such as glucose and sucrose.
April 29, 2024
15 minutes