Sialylation of Immunoglobulin E Is a Determinant of Allergic Pathogenicity

Immunoglobulin E (IgE) is a key molecule in allergies - the over-reaction of our immune system to non-harmful antigens from our surroundings. Upon sensitization, which happens at initial contact with antigen (in this case – allergen), allergen-specific IgE is synthesized and sits on the surface of cells (mastocytes an…

Immunoglobulin E (IgE) is a key molecule in allergies - the over-reaction of our immune system to non-harmful antigens from our surroundings. Upon sensitization, which happens at initial contact with antigen (in this case – allergen), allergen-specific IgE is synthesized and sits on the surface of cells (mastocytes and basophils) bound to a high-affinity receptor FcεRI. Following the next exposure, the allergen binds to these IgE molecules, crosslinking the FcεRI on the cell surface and triggering the immune cascade which results in a well-known spectrum of symptoms, ranging from mild runny nose and itching to life-threatening anaphylactic shock.
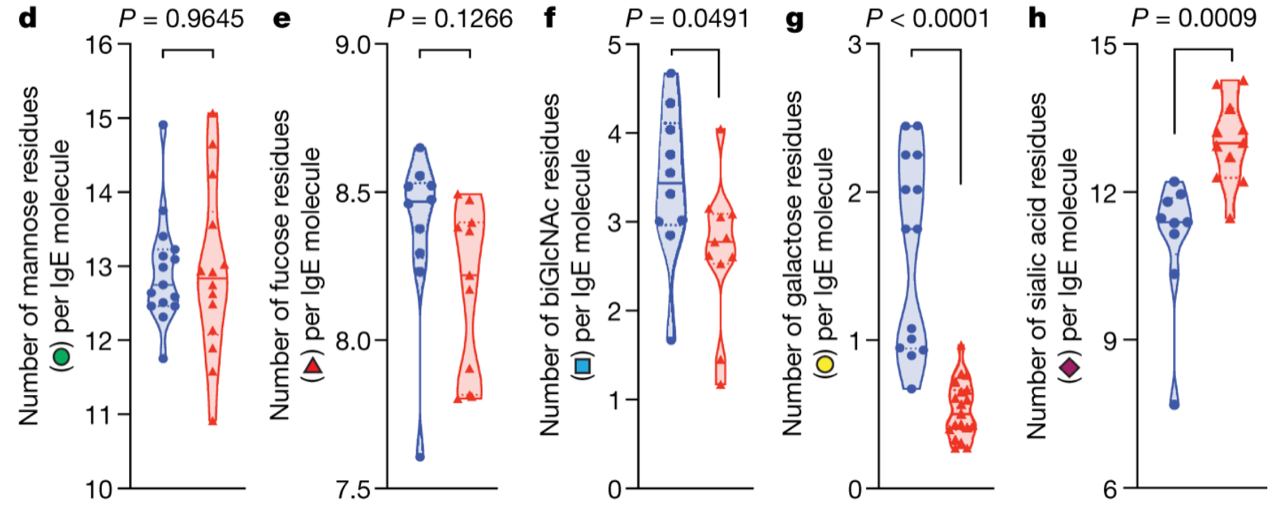
Total and allergen-specific IgE concentration in serum is routinely used to assess patients’ sensitization status. However, these measurements don’t accurately correlate with an allergic response. In the new Nature paper by the Anthony group at Harvard, we learn that the secret is in IgE glycans: IgE glycan composition and specifically sialylation is established as an important regulator of allergic disease.

Turn health content into action
Explore how biological age testing reflects immune health and inflammaging, GlycanAge for personal wellness tracking, and how to order a GlycanAge biological age test.

The Future of Healthcare is Preventative, Personalised, and Powered by Glycans
Whether you’re improving your own health, supporting patients, or driving research, GlycanAge helps you turn science into action.

How Living Against Your Body Clock Can Compromise Your Immune System
Some people fall asleep within minutes, anywhere, in any position. Others need a carefully constructed evening routine, the right temperature, the right level of quiet, and still end up lying awake for hours. It is easy to assume that the first group has nothing to worry about when it comes to sleep.
April 7, 2026
15 minutes

How Insulin Resistance Can Accelerate Aging Before Diabetes Ever Appears
When most people hear "insulin resistance", chances are their mind goes straight to a scary place: diabetes. And that makes sense, because that's almost always the context it appears in. But framing insulin resistance purely as a diabetes risk misses something important: long before it ever becomes diabetes, insulin r…
March 23, 2026
10 minutes