Antibodies are one of the main weapons in our arsenal for the eternal war against pathogens. They are an elaborate tool that can specifically recognise any foreign structure in our body. After binging to a foreign object, antibodies have to activate proper molecular mechanisms to deal with this foreign, non-self, object. If this non-self antigen is a pathogenic virus or a bacterium, it has to be eliminated in the most efficient way. However, if this foreign antigen is a food we eat, dust, or some being antigen in the air, then this antigen should be ignored, and activation of the immune system should be avoided.
A big part of this decision process is executed by the addition of different glycans to antibodies in a process called alternative glycosylation. This enables flexible and dynamic regulation of antibody function and is extensively used to fine-tune our immune system. Dozens of genes work together in this complex process that is also affected by many other factors, including hormones.
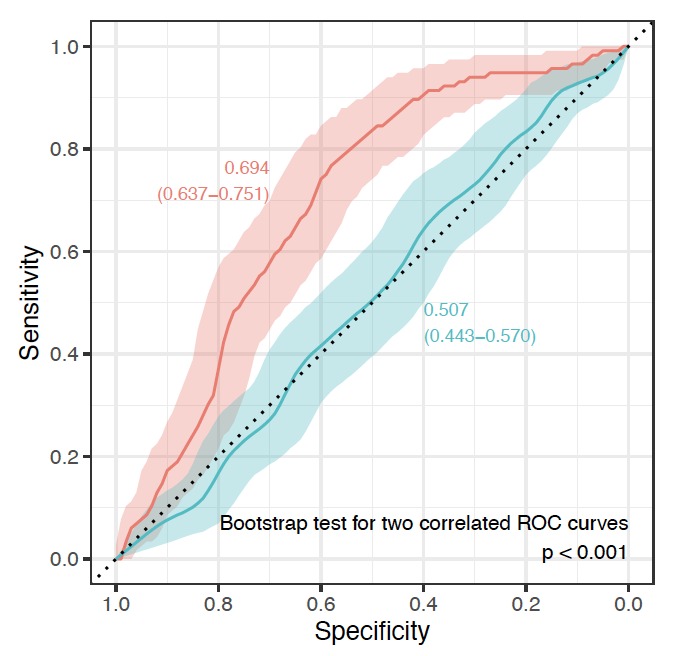
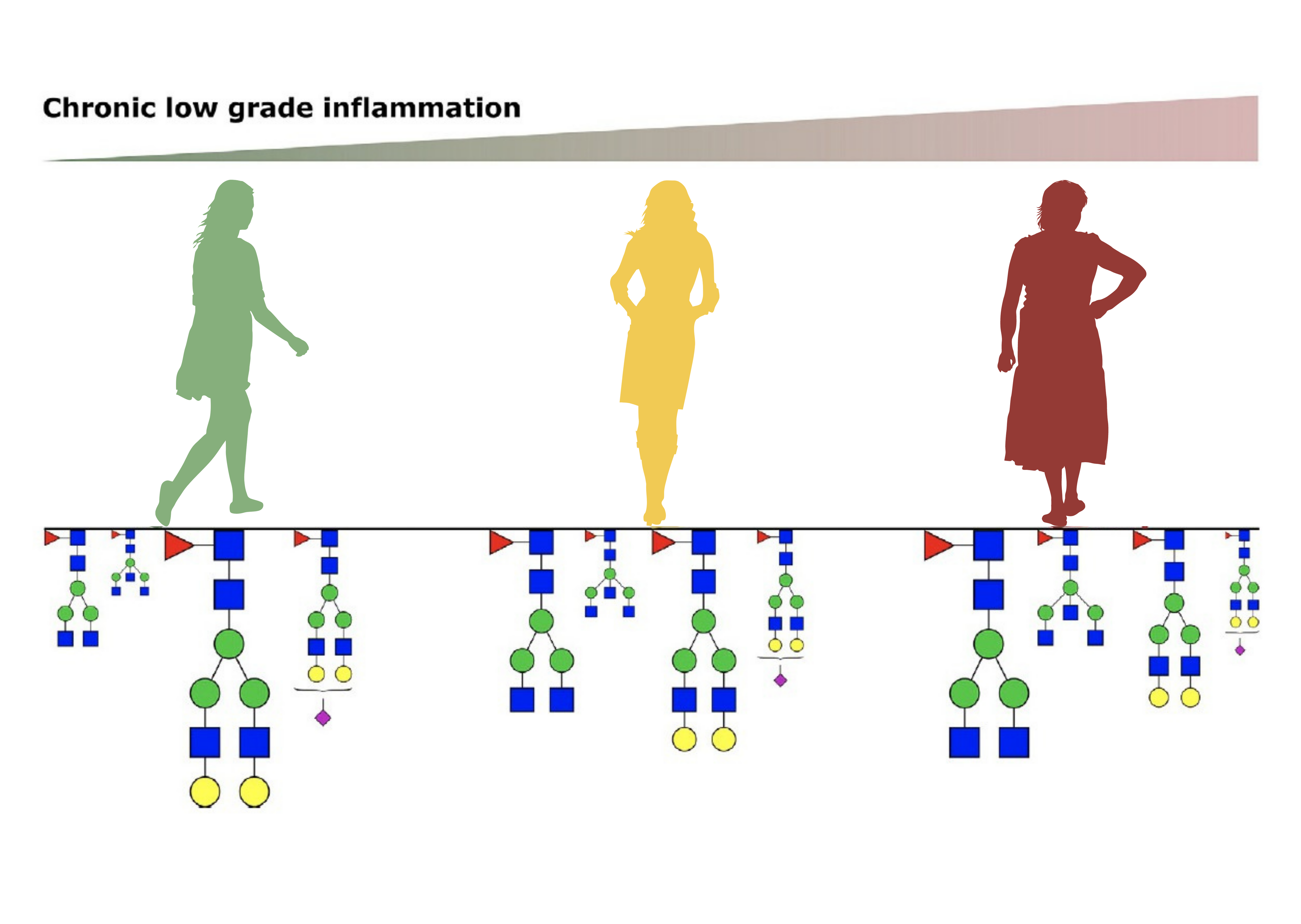
By analysing IgG glycome in 1,940 female twins at multiple time points researchers discovered that ageing of the IgG glycome is particularly prominent during the perimenopause when women transition from regular hormonal cycles to menopause. This is just an initial discovery, but it calls for further research in this direction since changes in GlycanAge glycome and consequential increase in low-grade chronic inflammation may be one of the molecular mechanisms behind increased health risks associated with menopause.
https://www.medrxiv.org/content/10.1101/2021.04.10.21255252v1
Other articles you may like:
 Glycoscience
GlycoscienceA Roadmap to Future
The Sustainable Development Goals or Global Goals are a collection of 17 interlinked goals designed to be a "blueprint to achieve a better and more sustainable future for all". The SDGs were set in 2015 by the United Nations General Assembly and are intended to be achieved by the year 2030. Glycoscience plays a role in achieving eight of the seventeen.

By Ph.D. Marina Martinic Kavur
 Glycoscience
GlycosciencePredicting Vulnerability to Stress Using Glycans
Can our glycans tell us something about our vulnerability to stress?

By Thomas Klarić
 Glycoscience
Glycoscience




 By Ph.D. Marina Martinic Kavur
By Ph.D. Marina Martinic Kavur
 By Thomas Klarić
By Thomas Klarić